Do Pfizer documents prove the COVID-19 vaccine claim of 95% efficacy is "completely false"? No, that's not true. It's also not true that the vaccine hurts fetuses: While the 95% efficacy claim cited in memes on social media is a small exaggeration of actual study results, Pfizer documents submitted for emergency use authorization (EUA) and licensure of the vaccine showed it was at least "91% effective in preventing COVID-19 disease," the U.S. Food and Drug Administration (FDA) said. Both the FDA and Pfizer say the vaccine has so far proven to be safe and effective for pregnant mothers and their unborn children.
The claims appeared in an Instagram post on May 6, 2022, titled "They lied and we were right. The truth will always prevail." It opens:
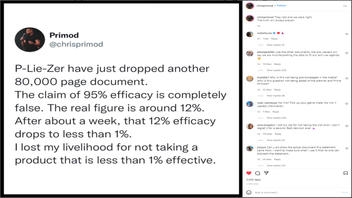
P-Lie-Zer have just dropped another 80,000 page document.
The claim of 95% efficacy is completely false. The real figure is around 12%.
After about a week, that 12% efficacy drops to less than 1%.
I lost my livelihood for not taking a product that is less than 1% effective.
This is what the post looked like on Instagram on May 9, 2022:
(Source: Instagram screenshot taken on Mon May 9 16:08:39 2022 UTC)
More memes
Similar versions of the claims circulated on social media and websites featuring memes. This one appeared on America's best pics and videos on May 5, 2022:
(Source: Instagram screenshot taken on Mon May 9 18:02:56 2022 UTC)
This one was also posted on May 5, 2022, to Facebook:
(Source: Facebook screenshot taken on Mon May 9 18:02:56 2022 UTC)
Lots of documents
The Pfizer documents referred to in the social media posts were released as part of an ongoing Freedom of Information Act lawsuit in which the drugmaker must release at least 55,000 pages of vaccine review documents each month. Details of the FDA's approval of the Pfizer COVID vaccine (also known as Comirnaty) are available on the agency's website. In a May 9, 2022, email to Lead Stories, the FDA said:
To date, FDA and CDC [Centers for Disease Control and Prevention] have not identified any safety signals for harm to fetuses following administration of the FDA-approved and authorized COVID-19 vaccines. As with all vaccines, including COVID-19 vaccines, we will continue to carefully monitor the vaccines for safety. For your information, CDC published a report in January from a study involving the Vaccine Safety Datalink. According to the report, in a large, multisite retrospective cohort study, receipt of COVID-19 vaccine during pregnancy was not associated with increased risk for preterm birth or small for-gestational age at birth. ... More info on this can be found in January's MMWR (Receipt of COVID-19 Vaccine During Pregnancy and Preterm or Small-for-Gestational-Age at Birth -- Eight Integrated Health Care Organizations, United States, December 15, 2020-July 22, 2021) available on CDC's website here.
With regard to the effectiveness of Comirnaty, in the data submitted by Pfizer from clinical trials in support of licensure (approval), the vaccine was 91% effective in preventing COVID-19 disease, with 77 cases of COVID-19 occurring in the vaccine group and 833 COVID-19 cases in the placebo group. For additional information about the data supporting licensure, please see the Summary Basis for Regulatory Action for the vaccine.
In a May 6, 2022, email to Lead Stories, Pfizer Media Relations said this about the company's COVID shot:
The vaccine has been shown to be safe and effective for pregnant mothers and is recommended by global health agencies around the world, such as the World Health Organization and the U.S. Centers for Disease Control and Prevention.
Numerous peer-reviewed studies and real world evidence have demonstrated that the Pfizer-BioNTech COVID-19 Vaccine is safe and effective. While clinical and real-world data still show that the Pfizer-BioNTech COVID-19 Vaccine provides a high level of protection against the virus, including all known variants, the companies are continuing to follow the science and exploring new vaccine approaches and regimens that may be needed as the pandemic evolves. We will continue to collect and assess all available data and remain in open dialogue with regulators and health authorities to help inform a COVID-19 vaccine strategy as the virus evolves.
With hundreds of millions of people vaccinated with the Pfizer-BioNTech COVID-19 vaccine, the safety profile and efficacy for the vaccine for all authorized groups continues to be favorable.
At the end of the vaccine's Phase 3 trials, it was determined to be "95% effective against COVID-19," a Pfizer press release on November 18, 2020, said. While the vaccine's efficacy rate against severe disease has dropped somewhat with the advent of new variants, modeling by the Imperial College London found that the Pfizer shot with a booster was around 85.9% effective against Omicron, compared with about 97% against Delta.
The CDC recommends the COVID vaccine for everyone 5 and above, including women who are pregnant or breastfeeding:
- COVID-19 vaccination is recommended for people who are pregnant, breastfeeding, trying to get pregnant now, or might become pregnant in the future.
- People who are pregnant should stay up to date with their COVID-19 vaccines, including getting a COVID-19 booster shot when it's time to get one.
- Evidence continues to build showing that COVID-19 vaccination during pregnancy is safe and effective.
Lead Stories previously debunked claims that COVID vaccines are harmful to pregnant women or their fetuses.
More than 12% effective
If Pfizer's COVID shot has a vaccine effectiveness (VE) rate of more than 80% even with the new variants, where did the 12% number come from in the memes? In a May 5, 2022, COVID Data Science blog, Jeffrey S. Morris, who holds a doctorate of statistics and who is the director of the Division of Biostatistics in the Department of Biostatistics, Epidemiology and Informatics at the University of Pennsylvania Perelman School of Medicine, said the misinformation appears to come from a Substack post by investigative journalist Sonia Elijah. He said her claims are based on the December 2020 Pfizer FDA briefing document for the Vaccines and Related Biological Products Advisory Committee (VRBPAC) meeting deciding the EUA on December 10, 2020:
It seems she is making the mistake of not realizing that 'suspected but unconfirmed cases' in the protocol refers to individuals receiving PCR tests because that had a symptom from a large pre-specifed list that triggered a PCR test, but for which the PCR test was negative. The authors of the FDA briefing document choosing the term 'suspected but unconfirmed cases' for those with negative PCR tests was a poor choice that has contributed to this confusion and led many to misinterpret what it means.
So the argument made by Sonia Elijah suggesting the 'real VE' of the Pfizer vaccines is 12% is an old one dating back to January 2021, and an erroneous one.