Did General Mills put a type of iron that is not able to be digested and used by the body in Cinnamon Toast Crunch? No, that's not true: the iron found in breakfast cereals is fine to consume and is digested by humans.
The claim appeared in a Facebook post (archived here) where it was published on April 22, 2021. The post contained five pictures of pure iron on a magnet. It opened:
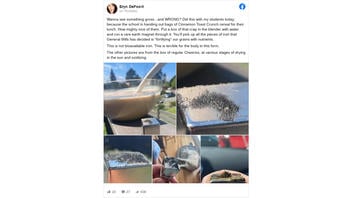
Wanna see something gross...and WRONG? Did this with my students today, because the school is handing out bags of Cinnamon Toast Crunch cereal for their lunch...This is not bioavailable iron. This is terrible for the body in this form."
This is what the post looked like on Facebook at the time of writing:
(Source: Facebook screenshot taken on Mon Apr 26 14:14:26 2021 UTC)
Bioavailable is defined by cancer.gov. as "the ability of a drug or other substance to be absorbed and used by the body."
Lead Stories reached out to General Mills, maker of Cinnamon Toast Crunch, for comment. Mike Siemienas, the brand media relations manager at General Mills, responded in an email on April 27 2021, saying, "The iron in our cereals is bioavailable and absorption in the body is enhanced by the presence of vitamin C, which we also fortify our cereals with. The FDA approves the use of the iron that we use to fortify our cereals." Iron, a key component in blood production, is listed by the FDA as an essential mineral.
It's the same iron that is found in the Earth's crust and part of our blood. On May 20, 2011, Scientific American published an experiment called, "Get the Iron out--of Your Breakfast Cereal" for students. The author writes about how the same iron found in our cereals is also found in many other places, including a multitude of foods, "Our bodies can't produce iron, but it's naturally present in many foods, including meats (beef, pork, turkey), produce (raisins, spinach, prunes) and nuts (walnuts, cashews, peanuts)." The author also notes that eating pure iron is not good for the body, so we need to get it in the digestible form that is found in food and vitamins.
The same Scientific American article has a step-by-step tutorial on how to do the experiment seen in the Facebook post.
In the conclusion of the Scientific American article, the author writes, "As it is Earth's rocks and soil, the iron in your breakfast cereal is attached to the other substances around it. But when you crush cereal down, it helps to free up the iron particles, so they can be picked up by the magnet (dissolving the other parts of the cereal frees up the iron even more)."
The Facebook post is correct in that the iron found on the magnet should not be consumed as it is shown, but if the iron had been left inside the cereal, it would've been fine to eat.