Do deaths reported to the Vaccine Adverse Event Reporting System (VAERS) associated with COVID-19 vaccinations all stem from 5% of vaccine batches? No, that's not true: There is no evidence of widespread issues with specific vaccine lots. Also, VAERS data do not establish a causal link between vaccine products and symptoms, making the relationship between the figures inconclusive on their own.
The claim appeared in a video (archived here) posted to Brand New Tube on November 2, 2021. The video is a clip from The Stew Peters Show, which features guest Jane Ruby. At the 0:42-mark of the video, Peters says:
A new analysis from a Daily Expose finds that of all of the deaths reported from COVID-19 vaccinations, 100% of them come from just 5% of vaccine batches. And even in that group, a handful of vaccine batches have had far more deaths and adverse reactions than the rest.
This is how the video looked on November 16, 2021:
The article cited in the claim can be found here on a website that makes claims that have been previously debunked by Lead Stories. Like the video making the claim, the article appears to suggest that data from VAERS, a database co-managed by the U.S. Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC), present a definitive link between reported symptoms and a vaccine product. But this is not true: according to the "About VAERS" page, the system is "passive":
VAERS accepts and analyzes reports of adverse events (possible side effects) after a person has received a vaccination. Anyone can report an adverse event to VAERS. Healthcare professionals are required to report certain adverse events and vaccine manufacturers are required to report all adverse events that come to their attention.
VAERS is a passive reporting system, meaning it relies on individuals to send in reports of their experiences to CDC and FDA. VAERS is not designed to determine if a vaccine caused a health problem, but is especially useful for detecting unusual or unexpected patterns of adverse event reporting that might indicate a possible safety problem with a vaccine. This way, VAERS can provide CDC and FDA with valuable information that additional work and evaluation is necessary to further assess a possible safety concern.
Therefore, VAERS reports offer no compelling evidence that the administration of COVID-19 vaccines is behind any deaths reported to VAERS.
In an email to Lead Stories on November 16, 2021, a spokesperson for the FDA further explained why the claim has no backing:
The assertion that all deaths come from only a small proportion of lots is not accurate. FDA's analysis of counts of serious events reported by lot does not show an unusual concentration of reports with a single lot or small group of lots. The number of reports submitted for a lot could vary based on important factors such as the lot size and length of time a lot has been in use ...
...As part of FDA and CDC's multi-system approach to vaccine safety monitoring, VAERS is designed to rapidly detect unusual or unexpected patterns of adverse events, also known as "safety signals." The claim you referenced incorrectly categorizes the VAERS reporting system. In general, a report made to VAERS does not mean that the vaccine, or other vaccines from the same group or lot, caused the event. If a safety signal is found in VAERS, further studies can be conducted in safety systems such as the CDC's Vaccine Safety Datalink (VSD) or the Clinical Immunization Safety Assessment (CISA) project. These systems do not have the same limitations as VAERS and can better assess health risks and possible connections between adverse events and a vaccine. Additional information about VAERS, VSD, and the CISA project are available here.
The FDA spokesperson explained the limitations of VAERS reports:
For example, a certain number of reports of serious illnesses or death do occur by chance alone among persons who have been recently vaccinated. Also, VAERS reports often lack important information, such as laboratory results used to help establish a potential association with the vaccine. For all serious events (life-threatening events, hospitalization, disability, death), follow-up with the health care provider and/or the parent or vaccinated individual is attempted in an in an effort to collect supplemental information on the reports. It is also important to point out that the sizes of vaccine lots vary and some are in distribution much longer than others. As such, a larger lot, or one that is in distribution longer, may be associated with more adverse events simply because more people have received vaccine from that lot. Additionally, while reports to VAERS often include a lot number, which is a useful and important aspect for safety surveillance, the reports can include inaccurately reported lot numbers.
The spokesperson added that the FDA and CDC have not identified a "safety signal" associated with a lot or group of lots. "Comparison of VAERS reports across lots should take into account assessment of lot size, time in use, and validity of the lot number," the spokesperson told Lead Stories.
The manufacturers of COVID vaccines also are involved in monitoring vaccine safety, according to the FDA spokesperson.
The manufacturers of the COVID-19 vaccines submitted pharmacovigilance plans to FDA to monitor the safety of their vaccines. The pharmacovigilance plan for each vaccine includes a plan to complete longer-term safety follow-up for participants enrolled in ongoing clinical trials. These plans also include other activities aimed at monitoring the safety of the vaccines to ensure that any safety concerns are identified and evaluated in a timely manner.
According to the CDC, if specific vaccine lots were deemed unsafe, they would typically undergo a recall initiated by the vaccine manufacturer:
There have been only a few vaccine recalls or withdrawals due to concerns about either how well the vaccine was working or about its safety. Several vaccine lots have been recalled in recent years because of a possible safety concern before anyone reported any injury. Rather, the manufacturer's quality testing noticed some irregularity in some vaccine vials. In these cases, the safety of these vaccines was monitored continuously before and after they were in use. CDC analyzed reports to the Vaccine Adverse Event Reporting System (VAERS) to search for any side effects that might have been caused by the irregularity, and found none. Any time such an irregularity is found in a vaccine lot which could make it unsafe, the manufacturer, in collaboration with the U.S. Food and Drug Administration (FDA), will recall it immediately. Information on recalled lots of is available by year from FDA.
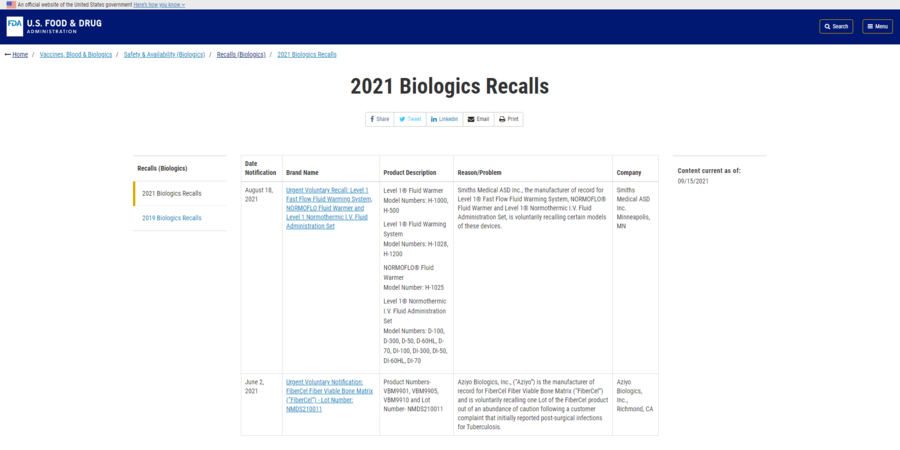
At the time of writing, no COVID-19 vaccines have been recalled nationally, as shown in the FDA "2021 Biologics Recalls" resource screenshotted below:
(Source: FDA screenshot taken on Mon Nov 15 14:49:26 2021 UTC)
Several vaccine lots are mentioned throughout the video, including Moderna COVID-19 vaccine lot 041L20A, which Ruby notes was briefly paused in California due to higher-than-average allergic reactions in the state. However, Ruby failed to acknowledge that after further testing, the lot was deemed safe for continued administration.
While administration of the Janssen COVID-19 vaccine was recommended for a pause by federal agencies, its administration also resumed after a safety examination. Lead Stories could not find evidence of any other COVID-19 vaccine or vaccine lot pauses in the U.S.
Lead Stories also reached out to the CDC, the U.S. Department of Health and Human Services, VAERS and COVID-19 vaccination manufacturers about this claim and will update this story with any relevant responses.