STORY UPDATED: check for updates below.
Does a Pfizer document show thousands of people around the world died from the company's COVID-19 vaccine the first month it was available? No, that's not true: The analysis of adverse event (AE) reports prepared by the pharmaceutical company doesn't determine cause and effect, only that more than a thousand vaccinated people died in the first three months after the vaccine's emergency use authorization from the Food and Drug Administration (FDA) on December 11, 2020.
Some 40% of the AE reports are "non-medically confirmed," according to the Pfizer document. Also, the meaning of the AE reports is unclear because not all the numbers are verified.
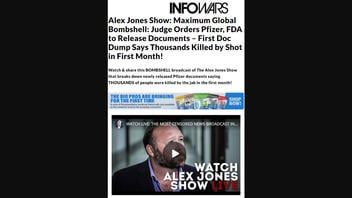
The claim appeared in an article (archived here) published by the InfoWars website on December 1, 2021, with the headline "Alex Jones Show: Maximum Global Bombshell: Judge Orders Pfizer, FDA to Release Documents - First Doc Dump Says Thousands Killed by Shot in First Month!" It opens:
Watch & share this BOMBSHELL broadcast of The Alex Jones Show that breaks down newly released Pfizer documents saying THOUSANDS of people were killed by the jab in the first month!
This is what the article looked like on December 2, 2021:
(Source: InfoWars website screenshot taken on Thu Dec 02 18:58:02 2021 UTC)
The Pfizer document was released as part of a Freedom of Information Act request by a group of more than 30 professors and scientists from universities including Harvard, Yale, Brown and UCLA. In their lawsuit filed September 16, 2021, the plaintiffs asked the FDA to share the data it relied upon in licensing Pfizer's COVID-19 vaccine.
The document, called 5.3.6 CUMULATIVE ANALYSIS OF POST-AUTHORIZATION ADVERSE EVENT REPORTS OF PF-07302048 (BNT162B2) RECEIVED THROUGH 28-FEB-2021, was approved on April 30, 2021, according to the timestamp on the side of the PDF. From December 1, 2020, through February 28, 2021, it notes 42,086 AE case reports with 1,223 fatal outcomes (see Table 1).
The number of deaths for those three months is less than the "thousands" claimed in the article for the first month of the vaccine alone. The document spells out its limitations:
Pfizer's safety database contains cases of AEs reported spontaneously to Pfizer, cases reported by the health authorities, cases published in the medical literature, cases from Pfizer-sponsored marketing programs, non-interventional studies, and cases of serious AEs reported from clinical studies regardless of causality assessment.
The limitations also include:
An accumulation of adverse event reports (AERs) does not necessarily indicate that a particular AE was caused by the drug; rather, the event may be due to an underlying disease or some other factor(s) such as past medical history or concomitant medication.
This is the breakdown of the 42,086 AE case reports by country:
Most cases (34,762) were received from United States (13,739), United Kingdom (13,404) Italy (2,578), Germany (1913), France (1506), Portugal (866) and Spain (756); the remaining 7,324 were distributed among 56 other countries.
In a December 3, 2021, email to Lead Stories, Keanna Ghazvini with Pfizer global media relations provided this statement from the company:
We take adverse events that are potentially associated with our COVID-19 vaccine very seriously. We closely monitor all such events and collect relevant information to share with global regulatory authorities. Based on ongoing safety reviews performed by Pfizer, BioNTech and health authorities, our vaccine retains a positive benefit-risk profile for the prevention of COVID-19 infections. To date, millions of people have been vaccinated with our vaccine. It is important to note that serious adverse events, including deaths that are unrelated to the vaccine, are unfortunately likely to occur at a similar rate as they would in the general population.
Most of the numbers reported in the Pfizer document come from expert groups and regulatory authorities, including European health organizations and the Vaccine Adverse Event Reporting System (VAERS) in the United States, which is run by the Centers for Disease Control and Prevention and the FDA.
Lead Stories has debunked several claims about vaccine deaths that misuse VAERS.
Anyone with internet access can add a report to the VAERS list of reports. The public access link to it expressly warns against unwarranted conclusions based on VAERS material because the list only provides a tally of unverified notes about any health event people experience after they are vaccinated.
The list itself cannot be used to prove or quantify, since all it shows is a chronological correlation, not the causal link that would be more difficult to establish. It's the equivalent of a police precinct's running "blotter" of reports that may serve as a starting point for police work, not an endpoint.
On December 13, 2021, the FDA responded to Lead Stories' request for a response to the InfoWars claim. In part, the agency said:
It's important to note that reports of adverse events to the Vaccine Adverse Event Reporting System (VAERS) following vaccination, including deaths, do not necessarily mean that a vaccine caused a health problem. In fact, reviews by FDA and CDC have determined that the vast majority of the deaths reported are not directly attributable to the vaccines. FDA requires healthcare providers to report any death after COVID-19 vaccination to VAERS, even if it's unclear whether the vaccine was the cause. Reports of adverse events to VAERS following vaccination, including deaths, do not necessarily mean that a vaccine caused a health problem.
Reports of death after COVID-19 vaccination are rare. As reported on CDC's website, more than 459 million doses of COVID-19 vaccines were administered in the United States from December 14, 2020, through November 29, 2021. During this time, VAERS received 10,128 reports of death (0.0022%) among people who received a COVID-19 vaccine.
Updates:
-
2021-12-15T21:14:21Z 2021-12-15T21:14:21Z Adds quote from FDA.