STORY UPDATED: check for updates below.
Does the regular use of ivermectin to prevent COVID-19 result in up to a 92 percent reduction in the disease's mortality rate and also reduce infection and hospitalization? No, that's not true: The claims about ivermectin were part of what one expert at the University of Nebraska Medical Center called "not a well-constructed study."
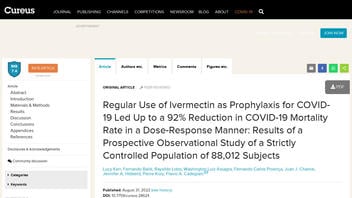
The claims appeared in a study (archived here) published by The Cureus Journal of Medical Science (Cureus) on August 31, 2022, under the title "Regular Use of Ivermectin as Prophylaxis for COVID-19 Led Up to a 92% Reduction in COVID-19 Mortality Rate in a Dose-Response Manner: Results of a Prospective Observational Study of a Strictly Controlled Population of 88,012 Subjects." It opened:
We have previously demonstrated that ivermectin used as prophylaxis for coronavirus disease 2019 (COVID-19), irrespective of the regularity, in a strictly controlled citywide program in Southern Brazil (Itajaí, Brazil), was associated with reductions in COVID-19 infection, hospitalization, and mortality rates. In this study, our objective was to determine if the regular use of ivermectin impacted the level of protection from COVID-19 and related outcomes, reinforcing the efficacy of ivermectin through the demonstration of a dose-response effect.
This is what the study looked like on The Cureus Journal of Medical Science website at the time of writing:
(Source: Cureus screenshot taken on Thu Sep 8 20:31:59 2022 UTC)
In a September 4, 2022, Twitter thread, University of Wollongong epidemiologist Gideon Meyerowitz-Katz tackled the study, everything from its title to its conclusions:
Let's look at the title first, because it's wrong. The authors describe this as a 'prospective observational study' of a 'strictly controlled population of 88,012 subjects'. That is incorrect
As the methods clearly states, the study design and ethics approval came AFTER the public health program occurred. This is more correctly termed a retrospective observational study of routinely recorded medical records
A National Library of Medicine (NLM) article says retrospective observational (cohort) studies have limitations:
Retrospective cohort studies, also known as historical cohort studies, are carried out at the present time and look to the past to examine medical events or outcomes. In other words, a cohort of subjects selected based on exposure status is chosen at the present time, and outcome data (i.e. disease status, event status), which was measured in the past, are reconstructed for analysis. The primary disadvantage of this study design is the limited control the investigator has over data collection.
The data for the study came from a citywide public health program that took place in Itajaí, Santa Catarina, Brazil, between July 7, 2020, and December 2, 2020. Participation and reporting in the program was voluntary.
Randomized controlled trials
Randomized controlled trials are considered the gold standard for effectiveness research, according to another NLM article, in this case, ivermectin as prophylaxis against COVID:
Randomized controlled trials (RCT) are prospective studies that measure the effectiveness of a new intervention or treatment. Although no study is likely on its own to prove causality, randomization reduces bias and provides a rigorous tool to examine cause-effect relationships between an intervention and outcome. This is because the act of randomization balances participant characteristics (both observed and unobserved) between the groups allowing attribution of any differences in outcome to the study intervention. This is not possible with any other study design.
In a September 9, 2022, email to Lead Stories, Dr. Stuart Ray, professor of medicine at the Johns Hopkins University School of Medicine, said the study sheds no new light on the use of ivermectin as a treatment for COVID:
The study is methodologically weak, making it hard to draw conclusions from its questionable results; however, we already have data from randomized controlled clinical trials that show no benefit of ivermectin for preventing severe COVID-19. In the context of those other more rigorous studies, and the flaws of this one, the report ... does not alter our understanding that ivermectin is ineffective for prevention or treatment of COVID-19 (SARS-CoV-2 infection related disease).
Dr. Kristin Mondy, chief of the Division of Infectious Diseases in the Department of Internal Medicine at The University of Texas at Austin Dell Medical School, said in a September 8, 2022, email that she sees the problems you would expect to find in a retrospective observational study:
After a read-through, I have main concerns with the statistical analyses. Not surprisingly, there was a huge difference in the type of people who took ivermectin (i.e. twice as many people over 50 took ivermectin compared to non-users). I don't really think that propensity-score matching can -- or should -- be used to overcome that degree of bias (hence a statistician should review).
I also think that studies based on self-reporting of medication without any additional checks on adherence are also prone to bias.
I'm honestly not all that familiar with this journal- I'm not sure how rigorous the peer review process is (or if it included a statistician).
Cureus identifies the study as being peer-reviewed. In a September 8, 2022, email to Lead Stores, Cureus Editor-in-Chief Dr. John R. Adler Jr., explained the journal's process:
Cureus requires a minimum of 2 completed reviews by independent reviewers. This article had 3 completed reviews and one partial review, with the latter providing a partial set of comments. This collection of reviews was available to the Cureus Approval Editor who must make the final decision to publish.
Cureus does not reveal the identities of the peer reviewers or their peer review comments, so it's uncertain whether a statistician reviewed the data as Mondy would recommend. In another September 8, 2022, email to Lead Stores, Cureus Director of Editorial Operations Graham Parker-Finger provided additional details:
Cureus conducts single-blinded peer review in which author names are divulged to reviewers while the reviewers themselves remain anonymous. ... Cureus requires all submitting authors to suggest a minimum of five potential peer reviewers, who are then vetted by our editors prior to the start of peer review.
In addition to these five reviewers, Cureus invites six reviewers with relevant domain expertise when the article enters peer review and three additional reviewers every two days until the article has satisfied peer review. These reviewers are drawn from our internal pool of reviewer volunteers and matched with articles based on specialty and keywords. In order to satisfy peer review, the article must receive at least two completed reviews from peer reviewers who were invited by the journal. In other words, any reviews submitted by author-suggested reviewers do not count towards the requirement of two completed reviews.
Cureus told Lead Stories it follows the International Committee of Medical Journal Editors' "Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals." The ICMJE website also lists Cureus among the journals saying they follow its recommendations. The ICMJE notes: "There may be some listed journals that do not follow all of the many recommendations and policies in the document."
In his September 6, 2022, email to Lead Stories, Dr. James Lawler with the Division of Infectious Diseases at the University of Nebraska Medical Center said he has several concerns about the study and whether it meets peer-review muster:
I think the first warning should be that it is published on an online portal that really isn't a traditional peer-reviewed scientific journal. ...
Several RCTs [randomized controlled trials] and legitimate meta-analyses by universally respected scientific groups like Cochrane Database have been done for ivermectin and the results are pretty clear - it provides no benefit in COVID-19.
Ray said he shares concerns about how painstaking the peer-review process was for this study:
This is a retrospective observational study in which comparisons are made in outcomes between people who may or may not have taken ivermectin. The accuracy of inferred ivermectin use seems to be low; this is illustrated by reports from the local health authorities in Brazil that many people took ivermectin who were not prescribed, and many who were prescribed ivermectin did not take it. In addition, people who developed COVID-19 were instructed to stop taking the drug, which could artificially strengthen the association between reduced use of ivermectin and COVID-19. These are examples, and reading the article gave me the impression that any peer review was not rigorous, possibly rushed.
Authors and conflicts of interest
Lead Stories has written about one of the study's authors before. Dr. Pierre Kory is a physician affiliated with the Front Line COVID-19 Critical Care Alliance (FLCCC), an anti-vaccine group that has been cited for perpetuating unsubstantiated claims regarding the virus in a scholarly article in Frontiers in Pharmacology. Kory is a founding member of FLCCC. Another of the study's authors, Dr. Flávio A. Cadegiani, is also a founding member of the alliance.
In the study's "Ethics Statement and Conflict of Interest Disclosures" section, the study's authors reveal their ties -- financial and otherwise -- to the FLCCC, an ivermectin manufacturer and ivermectin promotion groups.
Connections to drug manufacturers aren't unusual. Many peer-reviewed papers are backed by pharmaceutical companies. In his email to Lead Stories, Ray from Johns Hopkins said:
The Disclosures section of the article reveals that multiple authors including the lead author have financial or consulting relationships with a company that manufactures ivermectin and groups that have a long history of promoting its use. Such conflicts do not invalidate a study but they do represent potential conflict of interest, and the study's weak scientific methods don't mitigate these concerns.
For his part, Cadegiani provided this "post-publication message" in the comments section of the study on the Cureus website:
I cannot speak on the behalf of others.
However, while I received US$ 1,600 from an ivermectin manufacturer in the past, I consider it fair to clarify that:
1. This amount was far below from the time spent working on the consulting service.
2. The resulting consulting service was not positive for the molecule - payment was unrelated to the report I provided.
3. 100% of the amount received was donated for COVID-19 research and clinical management.
4. I have donated an equivalent of above US$ 200,000.00 for COVID-19.
5. I left a fully scheduled patients agenda to work on COVID-19 research projects for free.
Not FDA-approved
Asked for a response to the study's claims, Food and Drug Administration press officer Chanapa Tantibanchachai told Lead Stories in a September 6, 2022, email that the agency does not comment on third-party research:
Currently, FDA has not authorized or approved ivermectin for use in preventing or treating COVID-19 in humans. To date, published clinical trials have shown mixed results. Additional clinical trials assessing ivermectin tablets for the prevention or treatment of COVID-19 are ongoing. Information on clinical trials of ivermectin in COVID-19 can be accessed at https://www.clinicaltrials.
gov/. While FDA-approved products may be prescribed by physicians for unapproved uses if they determine it is appropriate for treating their patients, including during COVID-19, the safety and efficacy of ivermectin for the prevention or treatment of COVID-19 has not been established. Taking ivermectin can interact with other medications the patient is taking and cause side effects such as nausea, vomiting, diarrhea, hypotension (low blood pressure), allergic reactions (itching and hives), dizziness and ataxia (problems with balance).
The FDA offers a fact sheet discouraging the use of ivermectin against COVID-19: "Why You Should Not Use Ivermectin to Treat or Prevent COVID-19."
Updates:
-
2022-09-13T00:10:25Z 2022-09-13T00:10:25Z Removes reference to study being published on "open access" site.