Is water consumption so unhealthy that people "are not supposed" to drink it? No, that's not true: The claim on social media is an unsupported misrepresentation of the known facts about the mechanisms of hydration in the human body.
The claim originated from a video (archived here) published on TikTok on September 29, 2022, and which then resurfaced on social media in the summer of 2023. Its title said:
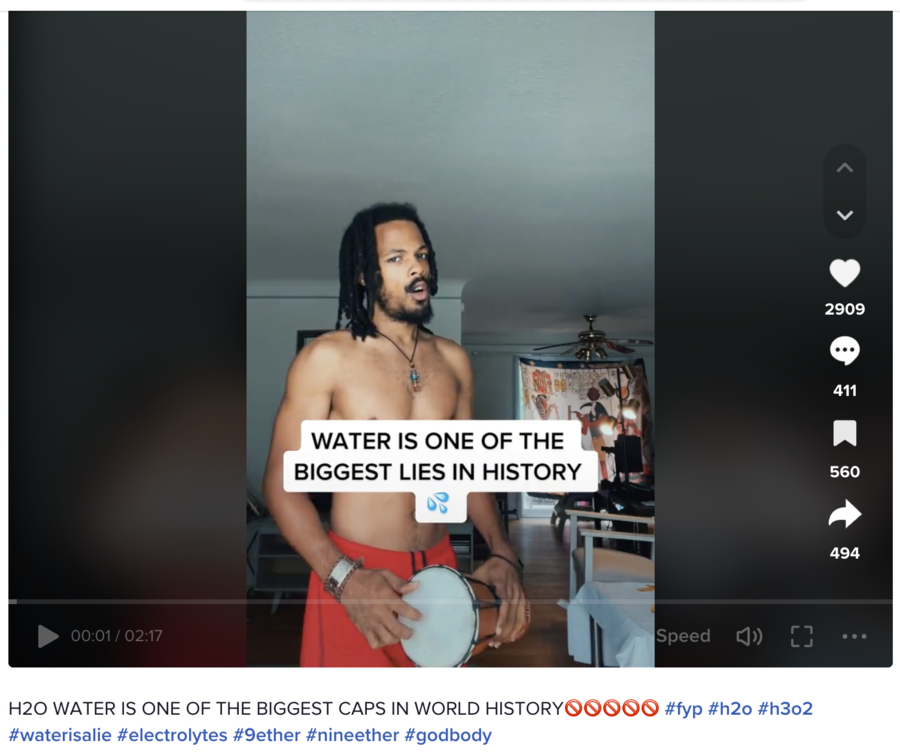
WATER IS ONE OF THE BIGGEST LIES IN HISTORY 💦
The man shown in the footage says at the 00:08 mark:
I want to tell you why you're not supposed to drink water. The water I'm talking about is H2O. That's the water we believe that we need, this same water, H2O, is the same water that we suspect is in our body. I'm going to break the news to you. H2O does not run through this body.
At the 00:53 mark, he continues:
When you do go to the hospital for dehydration, they put a needle in your arm, and they give you electrolytes via IV. They don't give it to you orally, they're not about to give you a bottle of water. They're gonna give you electrolytes through your arm, so we can hit your intracellular fluids and hydrate it. Not through a bottle of H2O, that's ridiculous! Why wouldn't they just give you water, if it's supposed to be the special ingredient? This is how hydration works -- hydration is the act of electrifying the cells. That's how hydration works. It needs electricity, potassium, magnesium, sodium, et cetera. It needs electrolytes! Water H2O is a dead mineral, it's not electrifying at all. Water is not hydrates you -- it actually dehydrates you!
The description below the video contained hashtags:
This is what the post looked like on TikTok at the time of writing:
(Source: TikTok screenshot taken on Thu Jun 8 19:38:17 2023 UTC)
The claim, however, distorts the science behind the process of hydration.
The website of Oregon State University explains that while minerals are important, water is also required with them:
The chemical reactions of life take place in aqueous solutions. The dissolved substances in a solution are called solutes. In the human body, solutes vary in different parts of the body, but may include proteins--including those that transport lipids, carbohydrates, and, very importantly, electrolytes...
In the body, water moves through semi-permeable membranes of cells and from one compartment of the body to another by a process called osmosis. Osmosis is basically the diffusion of water from regions of higher concentration to regions of lower concentration, along an osmotic gradient across a semi-permeable membrane. As a result, water will move into and out of cells and tissues, depending on the relative concentrations of the water and solutes found there. An appropriate balance of solutes inside and outside of cells must be maintained to ensure normal function.
The website of Colorado State University specifies that 80 percent of the initial process of water absorption occurs in the small intestine:
- Sodium is absorbed from the intestinal lumen by several mechanisms, most prominently by cotransport with glucose and amino acids, and by Na+/H+ exchange, both of which move sodium from the lumen into the enterocyte.
- Absorbed sodium is rapidly exported from the cell via sodium pumps - when a lot of sodium is entering the cell, a lot of sodium is pumped out of the cell, which establishes a high osmolarity in the small intercellular spaces between adjacent enterocytes.
- Water diffuses in response to the osmotic gradient established by sodium - in this case into the intercellular space. It seems that the bulk of the water absorption is transcellular, but some also diffuses through the tight junctions.
- Water, as well as sodium, then diffuses into capillary blood within the villus.
A study consisting of 72 male participants suggested that other drinks, including skim milk and orange juice, may get absorbed faster than still water, but it never made a case for a total exclusion of still water from one's diet.
While water does not contain electrolytes, another study that had hundreds of participants and five years of data reached the conclusion that most people don't have to consume sports drinks containing them: When one is low on salt and other minerals, people will naturally eat more food containing those elements to restore the conditions needed for proper water absorption.
A 2021 article published by one of the magazines of the American Physiological Society made the point that all liquids consumed during a day -- not only water but also tea, coffee, soups, fruits and the water in other types of food -- count toward reaching an optimal water balance in the human body.
While the idea that humans have to ensure the daily intake of a particular amount of water (for example, six or eight glasses) appears to be an exaggeration, the scientific community doesn't cast doubts on the vital role of hydration. The WHO, the CDC, the U.S. Department of Agriculture and the European Hydration Institute are among those credible organizations that emphasize the importance of access to clean drinking water and continue to recommend it to stay hydrated.
The website of Harvard Health Publishing, which is the consumer health education division of Harvard Medical School, summarizes some of the functions of the water you drink:
- carrying nutrients and oxygen to your cells
- flushing bacteria from your bladder
- aiding digestion
- preventing constipation
- normalizing blood pressure
- cushioning joints
- protecting organs and tissues
- regulating body temperature
- maintaining electrolyte (sodium) balance
The opposite -- dehydration -- may affect the work of the brain (which is one of the organs containing the largest amounts of water in the human body), kidneys, heart, skin, digestive system and overall thermoregulation.
IV fluids used to treat patients for dehydration in hospital settings are a faster way to deliver liquid into one's system. However, there are several types of fluids, and some of them might artificially elevate the concentration of minerals to the point when they may potentially start causing other complications in some people, reads a 2018 study published in the New England Journal of Medicine.
Another downside of hydration through IV fluids is that it doesn't appear to be the best long-term solution for healthy people because of the associated risks. As with any IV therapy, IV fluids may accidentally go into the tissues outside the vein, harming that tissue, or may result in swollen veins, which, in turn, may lead to blot clots. Additionally, if a bubble of air enters the blood through the needle, that may cause heart and breathing problems.
The H3O2 formula mentioned in hashtags below the post is only a temporary, "fractions-of-a-second" association in water that "has absolutely no effect" on its properties, as Lead Stories previously wrote. Politifact additionally clarified that H3O2 does not come from fruits.
Other Lead Stories fact checks about water-related claims can be found here.