Is a paper by Dr. Drew Pinsky, Dr. Peter McCullough, Dr. Harvey Risch, and nine others claiming that ivermectin and mebendazole improve cancer outcomes in 84.4% of cases a proper medical study? No, that's not true: Research experts told Lead Stories that the paper relies on an uncontrolled, self-reported survey with major bias, no independent verification of diagnoses or outcomes, and significant conflicts of interest. These flaws mean it cannot demonstrate that ivermectin or mebendazole improves cancer outcomes.
The claim appeared in a paper (archived here) published on Zenodo on April 7, 2026, titled "Real-World Clinical Outcomes of Ivermectin and Mebendazole in Cancer Patients: Results from a Prospective Observational Cohort." It opened:
Background: Drug repurposing offers a pathway to identify accessible, low-toxicity cancer therapies. Ivermectin and mebendazole have demonstrated multi-target anti-cancer activity in preclinical models, including the inhibition of cancer cell proliferation and the targeting of cancer stem cells. This paper evaluates real-world patient-reported outcomes, safety, and adherence in a cohort of cancer patients utilizing this combination protocol.
This is what a composite of images from the paper looked like at the time of writing:
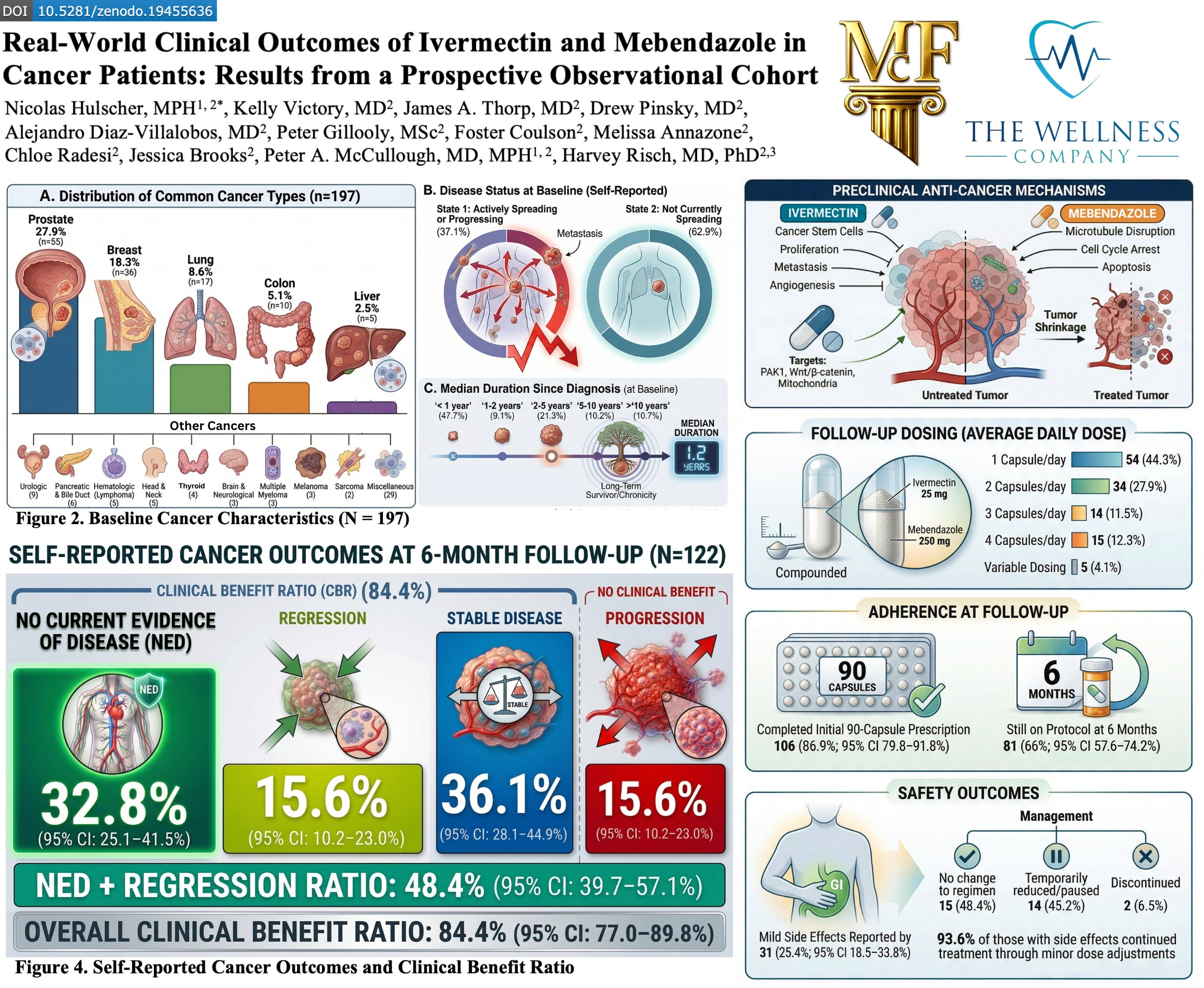
(Image source: Composite of figures from Real-World Clinical Outcomes of Ivermectin and Mebendazole in Cancer Patients: Results from a Prospective Observational Cohort)
This paper was not published in a peer-reviewed biomedical journal, as would be expected of a clinical trial claiming to show such a breakthrough in the treatment of cancer. Instead, it was published in Zenodo, a free, open-access, general-purpose research repository of work by researchers from all disciplines. There is no mention of peer review.
Peer review is crucial because it serves as a quality-control checkpoint, validating the accuracy, credibility, and originality of scholarly research before publication. Experts in the field scrutinize methods and conclusions, helping filter out invalid or low-quality work and ensuring scientific integrity. It also improves the clarity and rigor of research papers.
Conflict of interest
Gideon Meyerowitz-Katz, a senior research fellow at the University of Wollongong in Australia, said in an April 15, 2026, email that the study is very limited in its scope because it's a survey of patients attending a clinic run by the authors of the study:
This is a private clinic called The Wellness Company, which sells a variety of supplements and other non-standard treatments. All of the authors report being affiliated with this company and stand to financially benefit from the results of their publications. This is a very significant conflict of interest, which is appropriately reported in the study.
The "Conflict of Interest Statement" can be found on page 15 of the paper. Ivermectin and mebendazole are sold through the company website:
All authors are affiliated with and/or receive salary support from The Wellness Company (TWC), which operates the telemedicine platform through which the ivermectin-mebendazole combination evaluated in this analysis was prescribed and dispensed. TWC also offers compounded formulations of ivermectin and mebendazole as part of its clinical services.
Dr. Otto Yang, a professor of medicine and microbiology, immunology, and molecular genetics at the UCLA David Geffen School of Medicine, told Lead Stories in an April 14, 2026, email that the conflict of interest is concerning, but isn't a "fatal flaw" by itself:
But it does raise serious questions about objectivity and is a big red flag. It is also notable that the funding statement says that no 'external funding' supported this study; what was the internal funding? Presumably, it is TWC, which is highly suspect as well.
Methodological flaws
For Yang, the "fatal flaw" is found in the methodology of the trial used to collect the information. It's something, he said, the authors acknowledge in the "Conclusions" section of the paper (page 2).
When they say it is 'hypothesis-generating,' that is an acknowledgement that no conclusions can be drawn from the study, and that, at best, it raises questions.
Meyerowitz-Katz called the study "very minimal in scope." He continued:
The authors surveyed patients at entry to the clinic, at which point they were all prescribed ivermectin and mebendazole, and then again at 4-7 months after.
At baseline, 62.9% of the patients had cancer that was not actively spreading. It is hard to know precisely what proportion of these individuals were currently receiving treatment for their cancer, but the article does show that 42.1% had or were having surgery, 31.5% had or were having chemo, etc.
At follow-up, more than a third of the sample dropped out. Of the remainder, 48.4% reported no evidence of disease or regression.
What can be made of all these numbers? Not much, Meyerowitz-Katz said:
This provides us almost no usable information on the patients or their cancer. For one thing, there is no information on how these diagnoses were confirmed. The authors say that all patients had 'confirmed' cancer, but provide no details on how this confirmation was done. The surveys were anonymous, as the authors describe in their methodology. It is therefore unclear how we can be sure that these people had cancer.
The outcome measure is also of unclear relevance -- we do not generally use self-report to determine clinical efficacy in cancer treatments. More commonly, you'd use a scan or biopsy to confirm that the cancer had stopped progressing. This is also true for the determination of cancer in the first place -- this is the first time I've seen a diagnosis be established through an anonymous online survey.
Yang expressed similar concerns with the paper:
- All outcomes are self-reported. There is no verification of outcomes or that participants even took the prescribed ivermectin and mebendazole.
- There is huge recruiting bias. Patients who have political inclinations regarding ivermectin and mebendazole would be more likely to enroll, and therefore also be more likely to report benefit.
- There is huge reporting bias. Dead patients can't respond. Sick patients are less likely to respond. Patients doing well are more likely to respond.
- There is no control group who didn't receive ivermectin and mebendazole. Given that all patients are receiving cancer treatments, the good outcomes are likely to be from their conventional cancer treatment.
Their conclusions
In the end, both research experts dismissed the paper's claim of an 84.4% "clinical benefit" of ivermectin and mebendazole in the treatment of cancer, where "clinical benefit" refers to a positive, measurable impact on a patient's health -- specifically how they feel, function, or survive as a result of treatment or a medical device.
Meyerowitz-Katz concluded:
Overall, I would say that this paper provides no useful information about ivermectin, mebendazole, and cancer treatment. It is an anonymous survey conducted by people with a very significant financial conflict of interest, and mostly tells us that the patients who use their online clinic think that the clinic was useful. What this means for any clinical outcome is unclear.
Yang added:
All in all, this is total junk. The pretty pictures [illustrations in the paper] are for propaganda, not for scientific readers, and have little to no scientific content.
Department of Health and Human Services (HHS)
In the United States, the HHS plays a central role in drug approval through the Food and Drug Administration (FDA), which oversees the regulatory framework to ensure drugs are safe. Asked by Lead Stories about the benefits of ivermectin and mebendazole in the treatment of cancer, HHS Public Affairs responded in an April 10, 2026, email. It said:
Neither are currently FDA-approved for treating cancer.
Ivermectin tablets are approved by the FDA to treat people with intestinal strongyloidiasis and onchocerciasis, two conditions caused by parasitic worms. In addition, some topical forms of ivermectin are approved to treat external parasites like head lice and for skin conditions such as rosacea.
Mebendazole is approved to treat people with gastrointestinal infections caused by types of hookworms, roundworms, and whipworms.